About Lesson
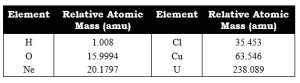
RELATIVE ATOMIC MASS
Relative atomic mass is the mass of an atom of an element as compared to the mass of an atom of carbon taken as 12.
Relative atomic masses are measured in atomic mass units.
1 a.m.u. is 1/12th part of mass of an atom of C-12.
1 a.m.u.= 1.661 x 10-27kg
= 1.661 10-24g
Examples: Na = 23 a.m.u.
Cl = 35.5 a.m.u